Research
Regulation of Cell Growth & Metabolism by Endocytosis
The Role of Sphingolipid Metabolism in Aging
Regulation of Cell Growth and Metabolism by Endocytosis
All cells must adapt to their surroundings to survive. Our research in the MacGurn Lab explores the core mechanisms behind this adaptive process, specifically focusing on how cells are remodeled via membrane trafficking and protein degradation. By understanding this fundamental biology, we hope to find new ways to combat disease. Some important fundamental questions that motivate our research include:
- How do cells sense nutrient availability and convert these signals into programs for growth or stress?
- How is transporter trafficking coordinated with cell signaling, metabolism, and growth decisions?
- How do endocytic processes interface with other complex cellular systems like metabolism and translation?
One lab project is focused on understanding how glucose transporters undergo regulated endocytosis. GLUT1 is responsible for glucose uptake in many human cells and tissues, yet very little is known about their regulation in a cellular context. Dissecting mechanisms that regulate GLUT1 subcellular localization and trafficking will have important implications for human diseases ranging from cancer to GLUT1 Deficiency Syndrome.
Recent Publication:
Qualls-Histed SJ, Nielsen CP, MacGurn JA. Lysosomal trafficking of the glucose transporter GLUT1 requires sequential regulation by TXNIP and ubiquitin. iScience. 2023 Feb 6;26(3):106150. doi: 10.1016/j.isci.2023.106150. eCollection 2023 Mar 17. PMID: 36890792.
Recent Preprint:
Hepowit NL, Singkhek HL, Frazier KD, Johnson DJ, MacGurn JA. Methionine-triggered growth arrest reveals activation of Gcn2 by methionine transporter endocytosis. bioRxiv. 2025. doi: 10.1101/2025.05.12.653625.
The Role of Sphingolipid Metabolism in Aging
- How does sphingolipid depletion increase longevity and healthspan in various aging models?
- How does sphingolipid composition and subcellular distribution change in aging cells and tissues?
- What are the mechanisms coupling sphingolipid metabolism and mitochondria function?
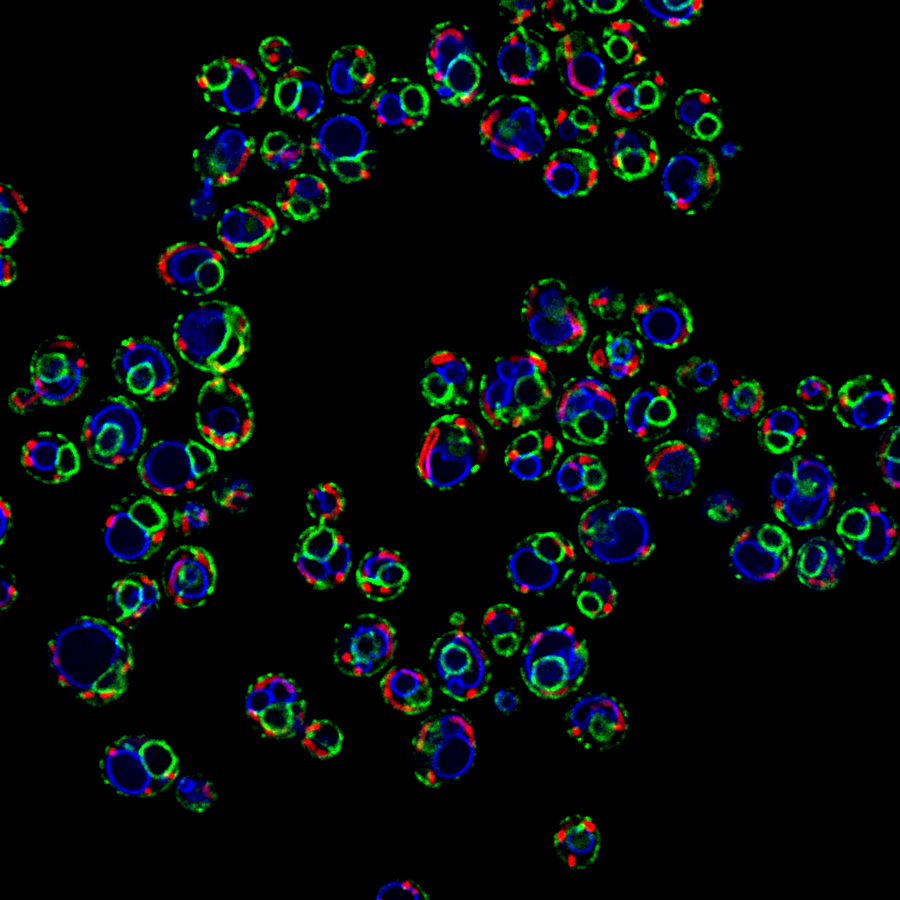
Recent Publication:
Hepowit NL, Moon B, Ebert AC, Dickson RC, MacGurn JA. Art2 mediates selective endocytosis of methionine transporters during adaptation to sphingolipid depletion. J Cell Sci. 2023 Jul 15;136(14):jcs260675. doi: 10.1242/jcs.260675. Epub 2023 Jul 25. PMID: 37337792.
Recent Preprint:
Ebert AC, Hepowit NL, Martinez TA, Vollmer H, Singkhek HL, Frazier KD, Kantejeva SA, Patel MR, MacGurn JA. Sphingolipid metabolism drives mitochondria remodeling during aging and oxidative stress. bioRxiv. 2025. doi: 10.1101/2025.02.26.640157.

